General Requirements, March 10, 2023
Export Procedures
- Export Procedures
- Transloading of Shell Eggs for Export
- Air Cargo Shipments
- LP-210S Online Exhibit (For exports to countries without a specification)
- LP-210S Stationary Lot Exhibit (For exports to countries without a specification)
- Disease-Free Statement – Other Countries (For export to countries without a specified DF Statement)
Ineligible Products
For information on restrictions on exports to specific countries, please consult the Shell Egg Export Restrictions Dashboard
Export Procedures - General Requirements March 10, 2023
Purpose
This instruction outlines general certification requirements for shell eggs exported from the United States. Shell egg exports certified by the Agricultural Marketing Service (AMS) must meet at minimum all the requirements of this instruction, in addition to country specific negotiated instructions.
Policy
- Establishment Responsibilities
Prior to processing, plant management is responsible for establishing detailed written procedures/record to provide for flock(s) identification and to maintain the identity of the shell eggs from the time of production through transport, storage, processing and packaging. Traceability is the responsibility of the applicant.
Plant management for the packaging facility must notify the USDA Grader of pending export orders, each certification request, and provide the Grader with the following information:
- Name and address of the producer
- Date of lay
- Flock identification and location information
- Size of lot to be certified
- If applicable, date product will arrive for processing
- When product is to be graded and shipped
- Other applicable documents referenced above prior to issuance of the export certificates.
- Agricultural Marketing Service (AMS) Biosecurity policy
- AMS employment policy prohibits personnel from owning avian species or operating farm with avian species.
- AMS personnel must follow each egg producer’s/packer’s established biosecurity procedure to prevent the risk of disease transmission.
- AMS and APHIS Agreement – Animal Health Certification for Shell Eggs
- AMS, Livestock and Poultry Program (LP) is recognized by the Animal and Plant Health Inspection Services (APHIS), USDA, as an authorized certifying agent.
- APHIS officials authorize animal health attestations for inclusions in AMS Certification Statements for Shell Eggs.
- APHIS officials notify AMS when any statement is affected, or animal health restrictions are imposed upon an identified geographical zone in the United States.
- Prior to certifying shell eggs for export, AMS Graders shall verify the origin processing facility and any source flock locations are not listed on the Shell Egg Export Restrictions Dashboard.
General Procedures
- Product Requirements
- All shell eggs must originate from chicken laying flocks (Gallus Domesticus) located in the United States of America and meet the criteria for current production, no more than 21 days old.
- Product must be processed and packed under continuous USDA supervision in an official USDA approved shell egg facility.
- Product must be washed and sanitized with compounds approved by USDA for food use.
- The shell eggs must be processed, packaged, labeled, and certified fit for human consumption in accordance with the Regulations Governing the Voluntary Grading of Shell Eggs (7 CFR Part 56).
- Grade Requirements
Unless otherwise stated within country-specific requirements, the shell eggs must be graded and certified as U.S. Grade B or better as outlined in the United States Standards, Grades, and Weight Classes for Shell Eggs (AMS 56).
- Animal Health Verification
Shell eggs must be derived from breeder flock(s) that participate in the National Poultry Improvement Plan (NPIP). The layer flocks must also be registered with the NPIP H5/H7 Avian Influenza Monitored Program.
- NPIP Breeder Flock Requirement: Producers must provide a copy(s) of APHIS Form VS 9-3 (Exhibit I), Report of Sales of Hatching Eggs, Chicks and Poults to the USDA Grader for each layer flock used to complete the export consignment. For intrastate movement, where the Form VS 9-3 would not be issued, producers must provide the USDA Grader with an invoice(s) listing their NPIP approval number for each flock. http://www.poultryimprovement.org/default.cfm
- NPIP Table Egg Layer Flock Requirement: Prior to processing, USDA Graders will verify that each source flock is listed on the NPIP website (pdf) for monitoring Avian Influenza. Eligible source flock(s) utilized to fill export orders will be recorded on the daily grading worksheet, LP-75 or LP-211.
http://www.poultryimprovement.org/documents/ShellEggLayerFlocksApprovedforExport.pdf
- Prevention of Salmonella Enteritidis in Shell Eggs during Production, Storage, and Transport
Shell egg producers must be in compliance with the Food and Drug Administrations (FDA), Final Rule for the Prevention of Salmonella Enteritidis in Shell Eggs during Production, Storage, and Transport (21 CFR 118). A producer receiving a “Letter of Warning” from FDA officials is not eligible to export until FDA provides information that corrective action(s) has been implemented. The LP National Office will notify appropriate regional staff when a facility is ineligible to export due to FDA regulatory action.
- Packaging/Packing
- All packing material must be new, showing no evidence of previous use.
- The interior packaging material (cartons and flats) must be new, showing no evidence of adhering foreign material or previous use.
- Pallets must be new or good used, free of visible evidence of adhering foreign material, and/or structural damage.
- Packing Case Labeling
- Each container must contain a safe handling or refrigeration statement, or be labeled ‘For Export Only’.
- Each primary container and shipping container must be identified with a statement of identity listed as ‘Eggs’, grade and size, official plant number and date of packaging.
- Each shipping case must be identified with the USDA Graded for Export Stamp (pre-printed on the shipping case label or traditional hand stamp) with the applicable certificate number.
- Labels must be printed or stamped in a central location on the end of each case. Handwritten or stenciled stamps are not acceptable.
- Retail Labeling
- It is the responsibility of the producer and exporter to understand the retail labeling requirements of the country of destination.
- AMS only certifies retail labeling when negotiated for specific countries of destination.
- Retail labeling not meeting FDA minimum labeling requirements must be specifically labeled ‘For Export Only’. Minimum FDA requirements include:
- Statement of identity
- A net contents statement
- Legal line such as ‘Packed by’ or ‘Distributed by’
- Safe handling instructions
- Nutritional facts panel
- Transport Unit
Transport units/containers used to export eggs from the origin facility must be of sound construction and clean. The refrigeration unit shall be set at 45°F or lower and is producing cool air before loading. Graders are to document the cleanliness of the transport unit and verification that the transport unit is supplied with functional refrigeration equipment on Form LP-75 or LP-211, as applicable. When unsanitary conditions are observed in a transport unit or the refrigeration equipment is not functional, the grader will notify a responsible plant official of the conditions. Product is not to be loaded on to the transport unit until the unsanitary condition(s) or function of equipment has been corrected. When such conditions occur, the USDA grader will document the unacceptable condition, including the name of the plant official notified, on the applicable worksheet.
The transport unit must be sealed (plant/company seal) prior to departure. The USDA grader is not required to be present at the time of shipment provided the condition of the transport unit is acceptable, the shipping cases are properly identified, and the seal(s) number is recorded on the certificate issued. The seal number must appear on the Form LP-210S certificate issued covering the shipment. If more than one container and/or seal is used from the time of loading at the processing facility until departure from a U.S. port, management must provide all seal numbers to the grader, and the grader must ensure these numbers are recorded on the Form LP-210S. It is the responsibility of the exporter to forward on the subsequent seals with the export documentation for application at the port.
- Transloading of Shell Eggs Certified for Export
In some cases, the final transport container may not be available at the location where the eggs are processed and certified. For example, small lots of eggs that will be added to other products at or near the port of departure, unavailable sea-van containers at the origin processing plant, etc. In order to assure that only sanitary containers are used for exported product, some countries request that the final shipping container be inspected by USDA personnel prior to loading of the product. For countries with specific transloading requirements, arrangements will be made, at company expense, to inspect the final transport container at a pre-determined location using the following guidelines:
- Origin Grader:
- Ensure all shipping cases have been stamped with the Graded For Export stamp containing the applicable certificate number.
- Complete the LP-210S showing the company seal number(s) that will be affixed to the final transport container and issue the certificate to plant management.
- Prepare Form LP-211 listing the seal(s) used on the truck trailer for shipment to the final inspection location.
- Provide plant management with the destination AMS Supervisor’s contact information.
- Fax or email a photocopy of the issued LP-211 to the destination Supervisor, advising that a shipment can be expected. Include the anticipated arrival date and details regarding the destination inspection location.
- Facility Management:
- Responsible for contacting the destination Supervisor’s office to arrange for a final sanitation inspection of the transport container.
- Assure that the issued LP-210S and company provided seal(s) is made available to the destination grader for final verification of shipping requirements.
- Destination Grader:
- Verify that the product arrived at port location with seals intact as indicated on Form LP-211. In the event that a shipment does not arrive as scheduled, or if seals are broken upon arrival, contact the Supervisor for further instructions.
- The final transport container will be inspected for cleanliness using the same criteria as listed above (Paragraph V. Transport Containers). Under no circumstances will product be loaded into a dirty container. It is not necessary to be present during the loading process as long as the company provided seal(s) are available and match the certificate, and the final transport container has been determined acceptable for cleanliness.
- Via email notification, the destination grader will advise the origin grader that the container condition examination was satisfactory. This communication will be filed with the export documents in the origin grader’s files.
- Grader’s time and expenses will be charged to the applicant requesting service using established billing procedures.
- Air Cargo Shipments
- Sealing of Air Shipments – Pallet and Container Sealing Procedures
Plant Management is responsible for informing the AMS Grader whether overseas export shipments will be shipped via sea-van container or by air. When requesting certification of export shipments to be transported via air freight, management shall provide a Certificate of Conformance to the AMS grader covering condition of the transport vessel. The CoC must be on company letterhead, signed and dated, containing the following information:- A statement that the product covered by Shell Egg Grading Certificate SEA XXXXXXX was transported under refrigerated conditions.
- A statement that the product was transported in a container or vessel (cargo hold) that is clean.
The CoC may be provided by a broker if the product is being warehoused by a company other than the producer. Management is responsible for providing the signed CoC from the broker to the AMS Grader for filing.
- Sealing of Individual Pallets
Due to the nature of air shipments, it is not typically possible to seal the cargo hold as with sea-van containers. In lieu of sealing the hold, the following protocol shall be used to accommodate the sealing of individual pallets. These seals shall be applied at the origin plant while the AMS grader is present. The type of seal must be designed so that pallets can be uniquely identified and would indicate tampering if broken. Sequentially numbered patch seals and tamper-evident tape are applied to the exterior of the pallet after plastic wrap and corner supports are in place. One possible method of accomplishing this is attached as an image in this document, though other methods may be used if these requirements are met and approved by an AMS supervisor. The sequentially numbered patch seals shall be applied to all four sides of the pallet(s).- The AMS Grader will use the following statement in the remarks section of the LP-210S in place of the seal statement: Air cargo: the following seals were applied to individual pallets: 001 – XXX
- Sealing of Air Shipments – Pallet and Container Sealing Procedures
When the individual pallets are sealed and destined for air transport, it is not necessary to seal the truck at origin.

- Unit Load Devices
Unit Load Devices (ULDs) are small metal and/or canvas containers commonly used to load aircraft with luggage or other cargo. They are not traditionally used for shell egg shipments due to their size, however they are acceptable for smaller export shipments. The containers must be capable of being sealed with serially numbered tamper-evident security seals. The seal numbers shall be documented on the LP-210S in the remarks section. AMS personnel are not responsible for sealing shipments at airports. It is the responsibility of the exporter or authorized representatives to apply the seals at the airport or secondary facility with accurate numbers as listed on the LP-210S certificate.
- Duration of Quality (Expiration Dating)
Expiration dating is voluntary. If a prefix, EXP or Best By, is used with a duration of quality that exceeds U.S. domestic policy, the following disclaimer will be printed in the remarks section of the Shell Egg Grading Certificate, LP-210S:
“The expiration date declared on the product is the responsibility of the applicant.”
If exporters wish to use longer periods, from 90 up to 180 days, each shipping case label must bear the statement “As Declared by Exporter” in close proximity to the EXP Date:
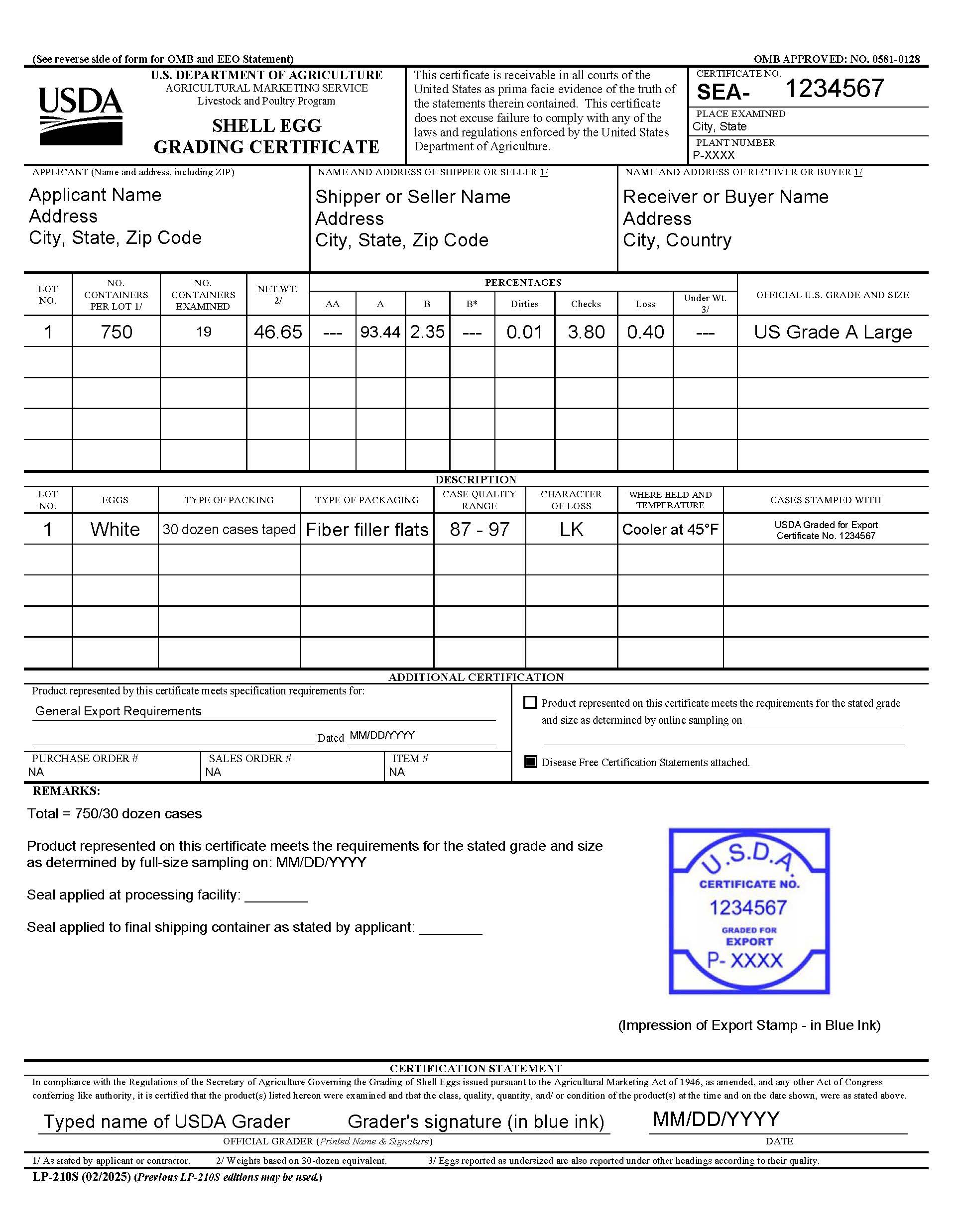
- Certification
The format of the export certificates shown in LP-210S exhibits for individual country specifications should not be changed beyond the inclusion of incidental information such as Purchase Order Numbers without approval from the National Office (Shell Egg). Graders cannot approve requests from applicants and brokers for significant changes to the remarks sections of export certificates to countries with approved specifications.
Numerous typographical errors may be justification by a foreign government border inspector to refuse entry of a shipment covered by the document. Graders should complete a thorough review of the certificate, Disease Free Statements, or any amendment before releasing the official export documents to plant management. Any requested corrections to export certificates should be issued using an official Amendment according to the USDA-LP certificate amendment policy, QAD 710.5b (pdf). A certificate superseding the original certificate cannot be issued without approval from the National Office (Shell Egg).
When completing the exporting documentation:
- All documentation must be typed.
- Applicant name and address must be recorded exactly the same as shown on the AMS Plant Query Site .
- Recipient address must be included in the buyer/receiver area. Indicating ‘to be sold’ on an export certificate is not acceptable.
- Grader name must be printed and signed.
- All signatures shall be in blue ink.
- The USDA grader must place the USDA Graded for Export stamp (in blue ink), which includes the sequential number of the certificate issued, on each official document [certificate, any amendment issued, and the Disease Free Statements].
- Seal number(s) must be included. If additional seals are included for sealing of final shipping container, seals shall be identified in the remarks section as “seal applied at processing facility” and “seal applied to final shipping container as stated by applicant.”
If the egg processor requests additional copies of the certificate for presentation to the purchaser(s) as an official document, a photocopy of the issued certificate may be signed (blue ballpoint ink pen) by the same grader issuing the certificate and the USDA Graded for Export stamp (in blue ink) placed on each requested document.
The exporter is responsible for presenting the original of these documents to authorities at the point of entry to the foreign country.